The Science of Editing Enzymes
APOBEC-3G and HIV Vif as new targets for drug discovery
- Miller, H.-J. Presnyak, V. & Smith, H.C. (2007) The dimerization domain of HIV-1 viral infectivity factor Vif is required to block virion incorporation of APOBEC3G. Retrovirology 4:81 PMC2222665.
- Salter, J.D., Morales, G.A. Smith, H.C. (2014) Structural Insights for HIV-1 Therapeutic Strategies Targeting Vif. Trends Biochem Sci. 39:373-80. PMC4511815
- Bennett, R.P., Stewart, R.A., Hogan, P., Ptak, R.G., Mankowski, M.K., Hartman, T.L., Buckheit, R.W., Jr., Snyder, B.A., Salter, J.D., Morales, G.A., Smith, H.C. (2016) An analog of camptothecin inactive against Topoisomerase I is broadly neutralizing of HIV-1 through inhibition of Vif-dependent APOBEC3G degradation. Antiviral Research 136:51-9 PMC5125868
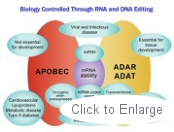
Editing enzymes are nucleotide deaminases that target select adenosines or cytidines within messenger RNA or within the DNA of select genes and change them effectively to guanosine or uridine. Editing of RNA and DNA changes ability of cells to code for specific proteins and as such is an important mechanism for regulating tissue function in both health and disease.
For example, editing enzyme function has been linked to our natural ability to fight off viral infection, central nervous system function, development of a robust immune system, regulation of ion transport through membranes and hormone receptor activity and risk reduction for atherosclerosis and cardiovascular diseases.
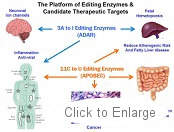
OyaGen’s technology is based on the platform of editing enzymes. OyaGen seeks to develop novel therapeutics to regulate editing enzyme activity. Patents are issued or pending on all proposed therapies.
The editing enzyme hA3G (also known as CEM15 or APOBEC-3G) serves as our natural defense against HIV-1 infection by inducing C to U mutations in the viral genome as it is being replicated. This results in defective viral proteins and suppression of the infectivity of the HIV-1 virus. The virus can protect itself from hA3G with its own protein called Vif (viral infectivity factor), which interacts with hA3G and tricks our cells into degrading hA3G. As a consequence HIV-1 is able to evade our natural defense. OyaGen has an exclusive license covering its ‘lead’ compound that functions to inhibit the activity Vif. Our compound antagonizes Vif’s ability to induce the degradation of hA3G thereby allowing the enzyme to catastrophically mutate the HIV-1 genome and reduce infectivity.
Efficacy of VDA in peptide form has been demonstrated on live HIV-1 virus under controlled laboratory conditions. OyaGen’s compound suppressed the infectivity of HIV-1 in human cells by 1000-fold compared to untreated cells infected with virus.
hA3G-Vif interaction and the proprietary rights that OyaGen holds in this important new therapeutic target sets OyaGen apart from other companies currently involved in the HIV market. The interaction is one of the most important AIDS therapeutic approaches of this century. HIV is less likely to escape OyaGen VDA because the target involves Vif’s interaction with cellular hA3G (something the virus requires in order to survive our defense system) affording the virus options of freedom through its inherent mutation frequency.
For additional information on editing enzymes see Dr. Harold Smith’s Lab Page.
Also see OyaGen on Wikipedia.
Platform Technology